By Mike Lesiecki, Ph.D.
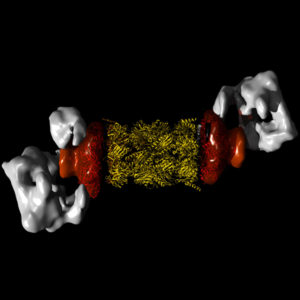
Until recently, the first step in the process of microscopy was to prepare the sample. For many electron microscopes you have to coat the samples with metal. This occurs in a vacuum and any water in, for example a biological tissue, is lost. But cell biology happens in water! This is where the cold (Cryo) part comes into play. The winners of 2017 Nobel Prize in Chemistry sparked a “New Era” in Biochemistry. Cryo-electron microscopes are used to “see the molecules inside the cell and how they interact.” Read more.
According to FEI, Cryo-Electron Microscopy (Cryo-EM) begins with vitrification, where the protein solution is cooled so rapidly that water molecules do not have time to crystalize, forming an amorphous solid that does little or no damage to the sample structure. The sample is then screened for particle concentration, distribution, and orientation. Next, a series of images is acquired, and two-dimensional classes are computationally extracted. In the final step, the data is processed by reconstruction software, yielding accurate, detailed, 3D models of intricate biological structures at the sub-cellular and molecular scales. These models can reveal interactions that were impossible to visualize previously, a key to scientific results.
Our webinar on January 26, 2018 focused on “Understanding the Biological Machinery by Cryo-EM.” Dewight Williams and Katia March, Research Scientists at the Leroy Eyring Center at Arizona State University presented. They work at the John M. Cowley Center for High Resolution Electron Microscopy and are part of the NCI-Southwest collaborative. You can view the recorded webinar here.
The technique is not for the faint of heart, the weak of pocketbook, or the absence of computational power. But the results can be amazing and insightful. Join us for our next NCI-Southwest webinar, March 23 on Protein Engineered Nano Materials.